|
The structures on the right side, with the OH group on the first carbon atom pointed upward, is the beta (β) form. The structure shown on the left side of Figure 2, with the OH group on the first carbon atom projected downward, represent what is called the alpha (α) form. When a straight-chain monosaccharide, such as any of the structures shown in ribose, forms a cyclic structure, the carbonyl oxygen atom may be pushed either up or down, giving rise to two stereoisomers. The anomeric carbon atom (colored orange here) is placed on the right. By convention for the D-family, the five-membered furanose ring is drawn in an edgewise projection with the ring oxygen positioned away from the viewer. Cyclic structures of this kind are termed furanose (five-membered) or pyranose (six-membered), reflecting the ring size relationship to the common heterocyclic compounds furan and pyran shown on the right.įor example, ribose, an important aldopentose, commonly adopts a furanose structure, as shown in the following illustration. These are the most important examples of cyclic hemiacetal formation in monosaccharides. This means that the aldehyde group in aldohexoses react with carbon 5 to format a six-membered ring, while in the case of aldopentoses, the aldehyde group in aldohexoses react with carbon 4 to format a five-membered ring. This carbon is now chiral, and it is called the anomeric carbon. Five and six-membered rings are favored over other ring sizes because of their low angle and eclipsing strain. The resulting structure will be an intramolecular cyclic hemiacetal. The carbonyl carbon (C1) becomes sp3 hybridized, with four different groups attached to it. In a monosaccharide, the carbonyl (C=O) and alcohol group (OH) exist within the same molecule, so they can react forming a cyclic hemiacetal (or hemiketal, in the case of ketoses). Image by Kupirijo at English Wikipedia, CC BY-SA 3.0, via Wikimedia Commons In organic chemistry, we described the reaction between carbonyl compounds and alcohol to form hemiacetal and hemiketal respectively, International Union of Pure and Applied Chemistry and International Union of Bio- chemistry and Molecular Biology, Joint Commission on Biochemical Nomenclature, Nomenclature of Carbohydrates (Recommendations 1996), Pure. Wiesner, K., “ The kinetics of mutarotation of D-Glucose with consideration of an intermediate free-aldehyde form”, J. E., “ Hemiacetal formation and the refractive indices and densities of mixtures of certain Alcohols and Aldehydes”, J. H., “ Ring-Chain Tautomerism of Hydroxy Aldehydes”, J. State of formaldehyde in the vapor phase”, Ind. 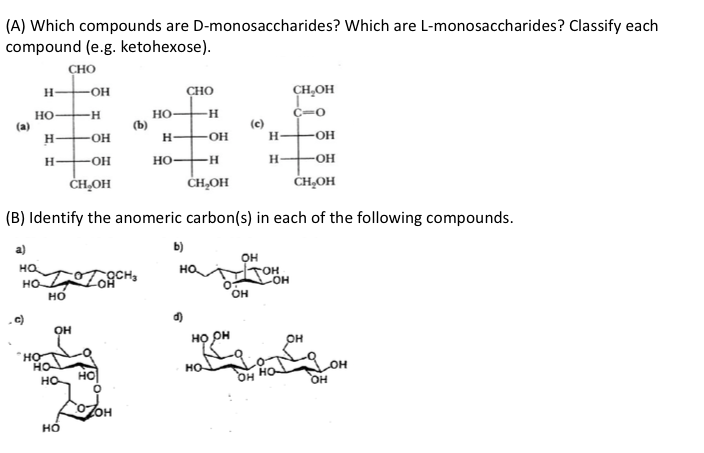
L., “ Distillation principles of formaldehyde solutions. N., The Constitution of the Sugars, Arnold, London, 1929 Tollens, B., “ Über das Verhalten der Dexrose zu ammoniakalischer Silberlösung”, Berichte (1883) 16, 921–924 Tanret, C., “ Molecular modifications of glucose”, Compt. Alexander-Jackson, E., “Rapid method of preparing Schiff’s reagent for the Feulgen test”, Science (1950) 111, 13 Demonstration a free aldehyde group in certain aldoses”, Ind. C., “ Supersensitive Schiff’s aldehyde reagent. 
Koenigs, W., “ Über fünffach acetylierte Galactose und Dextrose”, Berichte (1889) 22, 2207–2213įischer, E., “ber die Verbindungen der Zucker mit Alkoholen und Ketonen”, Berichte (1895) 28, 1145–1167 
L., “ Stereochemistry of Carbon Compounds”, McGraw-Hill, New York, 1962įischer, E., “ Über die configuration des Traubenzuckers und seiner Isomeren.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
